BUILDING THE PERIODIC TABLE
As you have seen and hopefully have heard, we are working across all Foundations of Science classes to build our understanding of the Periodic Table - both literally and figuratively!
Each student adopted an element back in December. During the month of January each of them compiled element research into an online Smore presentation and built a Bohr Model, which is now hanging from the ceiling of Room 200 and the hallway outside.
This week, we have taken our understanding of the elements a step further, expanding our view to look at the entire Periodic Table.
For three straight class days, I asked each student to place a giant Post-It note over his or her element on the Periodic Table made from Theodore Gray's Photographic Card Deck of the Elements.
As you have seen and hopefully have heard, we are working across all Foundations of Science classes to build our understanding of the Periodic Table - both literally and figuratively!
Each student adopted an element back in December. During the month of January each of them compiled element research into an online Smore presentation and built a Bohr Model, which is now hanging from the ceiling of Room 200 and the hallway outside.
This week, we have taken our understanding of the elements a step further, expanding our view to look at the entire Periodic Table.
For three straight class days, I asked each student to place a giant Post-It note over his or her element on the Periodic Table made from Theodore Gray's Photographic Card Deck of the Elements.
I posted each day's final Periodic Table picture on Schoology so that the students could study it and respond with a post identifying the trends the day's Post Its showed, using their adopted elements as reference points on the Table.
ELEMENT FAMILIES: DIGITAL PRESENTATIONS
One of the things I love most about using technology in class is that it allows us to do things that we otherwise wouldn't be able to do.
If I didn't have a way to connect my classes through Schoology, we would only be able to study 30 elements at a time. Because I can - and did - put all 91 of my ninth grade students together in one big Schoology group, we now have 92 (including my adopted element, Hydrogen) of the 118 known elements represented.
Instead of working in isolation or just within their class period, students are now collaborating across my three classes to produce fully digital presentations about representative groups of elements - what I like to call "element families."
Each element family has its own discussion board on Schoology, and the students started this short, snowy week off by introducing themselves and their elements. They also shared the link to the Smore they made when they did their individual element research with the other members of their families, so that they could build on what we read in our textbook to develop a deeper understanding of the properties their families of elements share.
Today we spent our first day in the computer lab getting started on their family presentations - and the kids did an AWESOME job! It was really fun to see them work face-to-face and communicate virtually to research and problem solve today. They were patient and organized, and I can't wait to see their finished products!
The Alkali Metals, Boron Family, Carbon Family, Nitrogen Family, Oxygen Family, Halogens, Noble Gases, and all three Transition Metals groups have each chosen to make a Prezi to teach their classmates about the properties, trends, and individual elements in their families. The Lanthanide Series students are making a Weebly website (like this one - only way cooler!), the Actinides are collaborating on a shared PowerPoint, and the Alkaline Earth Metals are working together on a Smore.
I know I'm stretching many of them way out of their comfort zones - using technology, learning new tech tools, working in groups, and working in groups with kids who aren't even in the same class...all in one project. This is a great learning experience on so many levels, and when the students spend next week learning from each other - not by standing up in front of a PowerPoint presentation but by sharing an engaging, fully digital presentation - I know they'll see how valuable this was.
We will spend Monday finalizing digital presentations (due to me in final form by Monday at 9 p.m.), then Tuesday and Wednesday of next week watching the digital presentations to learn properties and trends for each group. Next Thursday and Friday will be wrap-up and review days. Our Unit 4: Periodic Table exam is tentatively (pending future weather disasters) scheduled for Tuesday, February 9.
GROUP COLLABORATION GRADES
True confession: I hated group projects in high school. True confession #2: I hated them more in college. True confession #3: I'm not a huge fan of group projects in the adult world either. True confession #4: I was (okay, I still am) the one who did the whole thing because I didn't trust the slackers in my group to do things correctly (or at all), and I wasn't going to let my grade (or credibility, or moral conscience) suffer because of them. There I said it. (Let's face it. No one is surprised by this. Everything about me screams Type A, I think. I am fooling no one.)
As a teacher, I have always found it to be really hard to evaluate individual contributions to group projects. You can do anonymous group surveys, but a lot of times students are either too hard on themselves or not honest enough about how much work each person did. And when students have to meet up on the weekends to get a group project done, well, I'm not there to see who's working and who's not.
I love using technology to help me objectively assess each group member's contribution to their team's project in order to arrive at a 30 point (major) collaboration grade. In addition to visually watching students work during our two class work days, I have already been able to track who's been communicating on time with their element family through Schoology, and I can monitor what each person adds to their Prezis, Weebly, PowerPoint, or Smore - all of which have me listed as a shared editor or administrator. Everything's time and date stamped, everything's transcribed for me to go back and read. It's beautiful, really. (And I've already given the kids the "You can't hide" speech.)
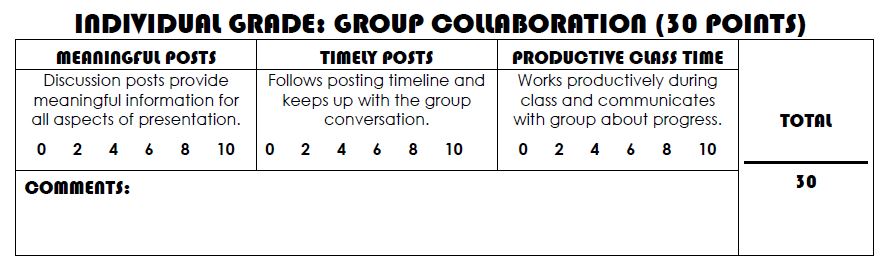
This is the rubric I am using for each of them. The students received a copy of this (on pink paper for easy locating) on Tuesday, January 19, when I introduced the project, and there is a digital version of it posted on the Resources page of our Schoology group.
ELEMENT FAMILIES: DIGITAL PRESENTATIONS
One of the things I love most about using technology in class is that it allows us to do things that we otherwise wouldn't be able to do.
If I didn't have a way to connect my classes through Schoology, we would only be able to study 30 elements at a time. Because I can - and did - put all 91 of my ninth grade students together in one big Schoology group, we now have 92 (including my adopted element, Hydrogen) of the 118 known elements represented.
Instead of working in isolation or just within their class period, students are now collaborating across my three classes to produce fully digital presentations about representative groups of elements - what I like to call "element families."
Each element family has its own discussion board on Schoology, and the students started this short, snowy week off by introducing themselves and their elements. They also shared the link to the Smore they made when they did their individual element research with the other members of their families, so that they could build on what we read in our textbook to develop a deeper understanding of the properties their families of elements share.
Today we spent our first day in the computer lab getting started on their family presentations - and the kids did an AWESOME job! It was really fun to see them work face-to-face and communicate virtually to research and problem solve today. They were patient and organized, and I can't wait to see their finished products!
The Alkali Metals, Boron Family, Carbon Family, Nitrogen Family, Oxygen Family, Halogens, Noble Gases, and all three Transition Metals groups have each chosen to make a Prezi to teach their classmates about the properties, trends, and individual elements in their families. The Lanthanide Series students are making a Weebly website (like this one - only way cooler!), the Actinides are collaborating on a shared PowerPoint, and the Alkaline Earth Metals are working together on a Smore.
I know I'm stretching many of them way out of their comfort zones - using technology, learning new tech tools, working in groups, and working in groups with kids who aren't even in the same class...all in one project. This is a great learning experience on so many levels, and when the students spend next week learning from each other - not by standing up in front of a PowerPoint presentation but by sharing an engaging, fully digital presentation - I know they'll see how valuable this was.
We will spend Monday finalizing digital presentations (due to me in final form by Monday at 9 p.m.), then Tuesday and Wednesday of next week watching the digital presentations to learn properties and trends for each group. Next Thursday and Friday will be wrap-up and review days. Our Unit 4: Periodic Table exam is tentatively (pending future weather disasters) scheduled for Tuesday, February 9.
GROUP COLLABORATION GRADES
True confession: I hated group projects in high school. True confession #2: I hated them more in college. True confession #3: I'm not a huge fan of group projects in the adult world either. True confession #4: I was (okay, I still am) the one who did the whole thing because I didn't trust the slackers in my group to do things correctly (or at all), and I wasn't going to let my grade (or credibility, or moral conscience) suffer because of them. There I said it. (Let's face it. No one is surprised by this. Everything about me screams Type A, I think. I am fooling no one.)
As a teacher, I have always found it to be really hard to evaluate individual contributions to group projects. You can do anonymous group surveys, but a lot of times students are either too hard on themselves or not honest enough about how much work each person did. And when students have to meet up on the weekends to get a group project done, well, I'm not there to see who's working and who's not.
I love using technology to help me objectively assess each group member's contribution to their team's project in order to arrive at a 30 point (major) collaboration grade. In addition to visually watching students work during our two class work days, I have already been able to track who's been communicating on time with their element family through Schoology, and I can monitor what each person adds to their Prezis, Weebly, PowerPoint, or Smore - all of which have me listed as a shared editor or administrator. Everything's time and date stamped, everything's transcribed for me to go back and read. It's beautiful, really. (And I've already given the kids the "You can't hide" speech.)
This is the rubric I am using for each of them. The students received a copy of this (on pink paper for easy locating) on Tuesday, January 19, when I introduced the project, and there is a digital version of it posted on the Resources page of our Schoology group.
I would love for you to have your student show you what they're working on with the members of their Element Families. Please don't hesitate to ask them for a sneak peek!
Have a wonderful weekend, and don't forget to look for Mercury, Venus, Saturn, Mars, and Jupiter lined up this week!
Have a wonderful weekend, and don't forget to look for Mercury, Venus, Saturn, Mars, and Jupiter lined up this week!